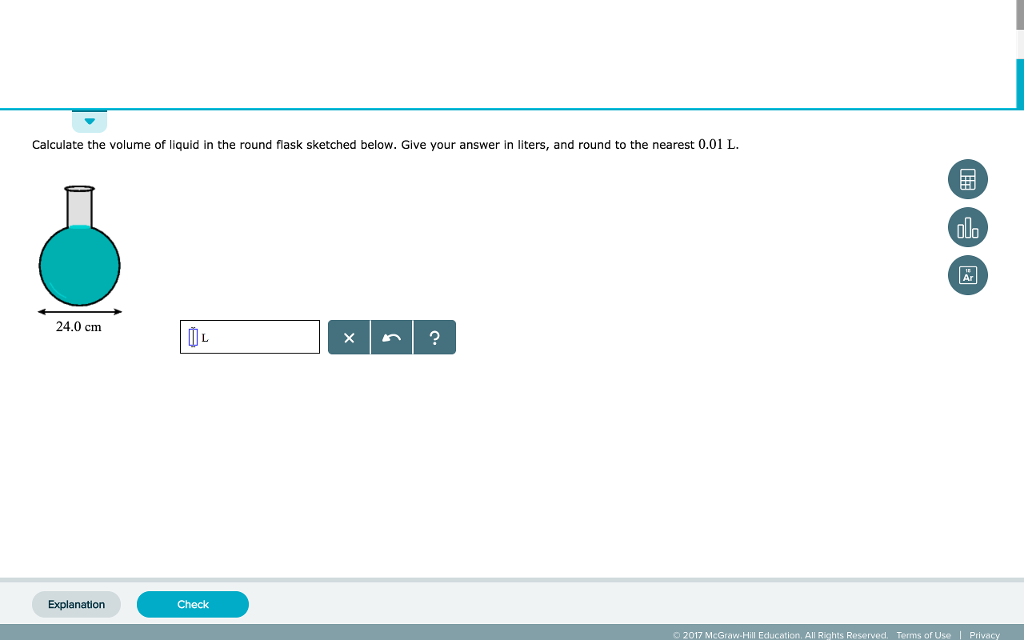
SOLVED:Finding the volume of a flask. A student obtained a clean dry glass-stoppered flask. She weighed the flask and stopper on an analytical balance and found the total mass to be 31.601
What is the volume of a conical flask which is 13 cm in height, with base radius of 6 cm, and an upper radius of 2 cm? - Quora

After completing this lesson you should be able to : Balanced equations show the mole ratio(s) of reactants and products. The molar volume is the same. - ppt download

A glass flask of volume one litre at 0^o C is filled level full of mercury at this temperature. The flask and mercury are now heated to 100^o C. How much mercury

A flask was heated from 27^(@)C to 227^(@)C at constant pressure. Calculate the volume of the flask if 0.1 dm^(3) of air measured at 27^(@)C was expelled from the flask.

SOLVED:Finding the volume of a flask. A student obtained a clean dry glass-stoppered flask. She weighed the flask and stopper on an analytical balance and found the total mass to be 31.601

Consider the flasks in the following diagrams: Assuming the connecting tube has negligible volume, predict what each diagram will look like once the stopcock between the two flasks is opened. Calculate the

SOLVED:An empty Erlenmeyer flask weighs 241.3 g. When filled with water (d=1.00 g / cm^3), the flask and its contents weigh 489.1 g . (a) What is the flask's volume? (b) How

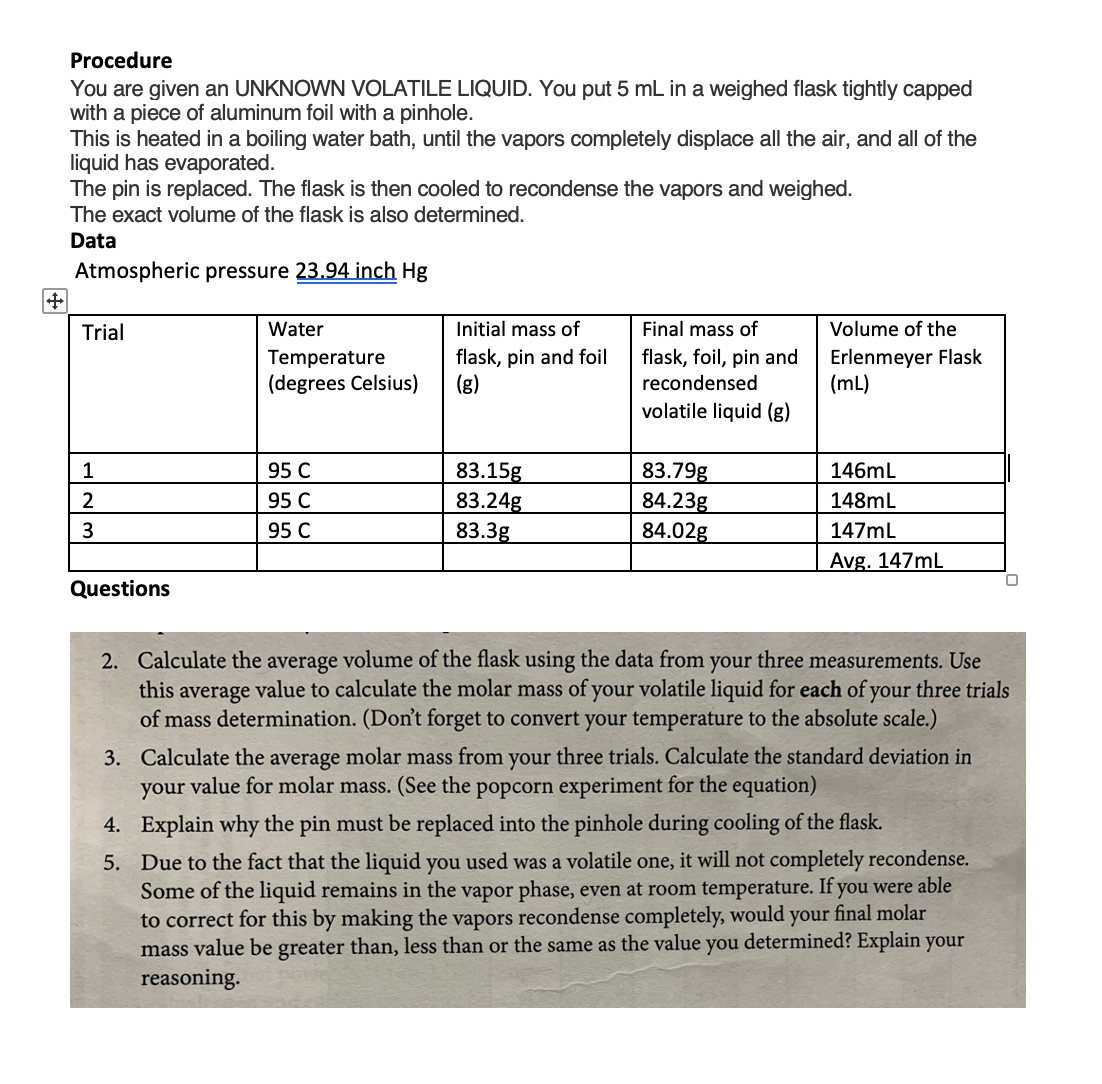
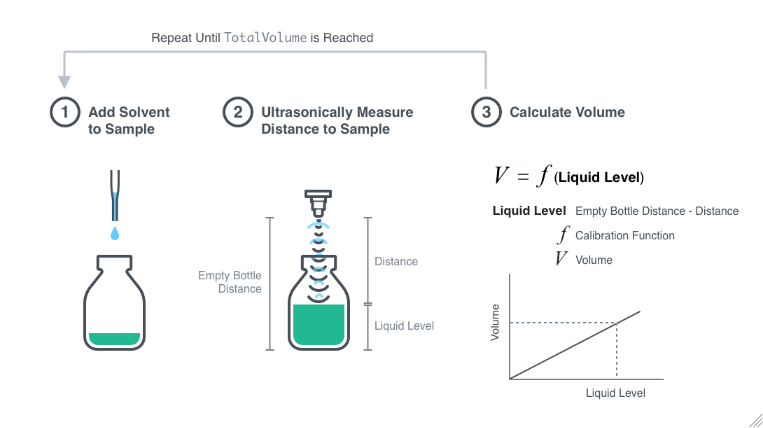
The volume of vapor in a flask can be determined by two methods: either by determining the mass of water required to just fill the flask completely or by using a graduated
0.01 mole of mehane and 0.96 g of oxygen were enclosed in a flask maintained at a temperature 300k.The pressure inside the flask was found to be 101325 nm.Calculate the volume of

Two flask of equal volume are connected by a narrow tube (of negligible volume) are at `27^(@)C` and - YouTube
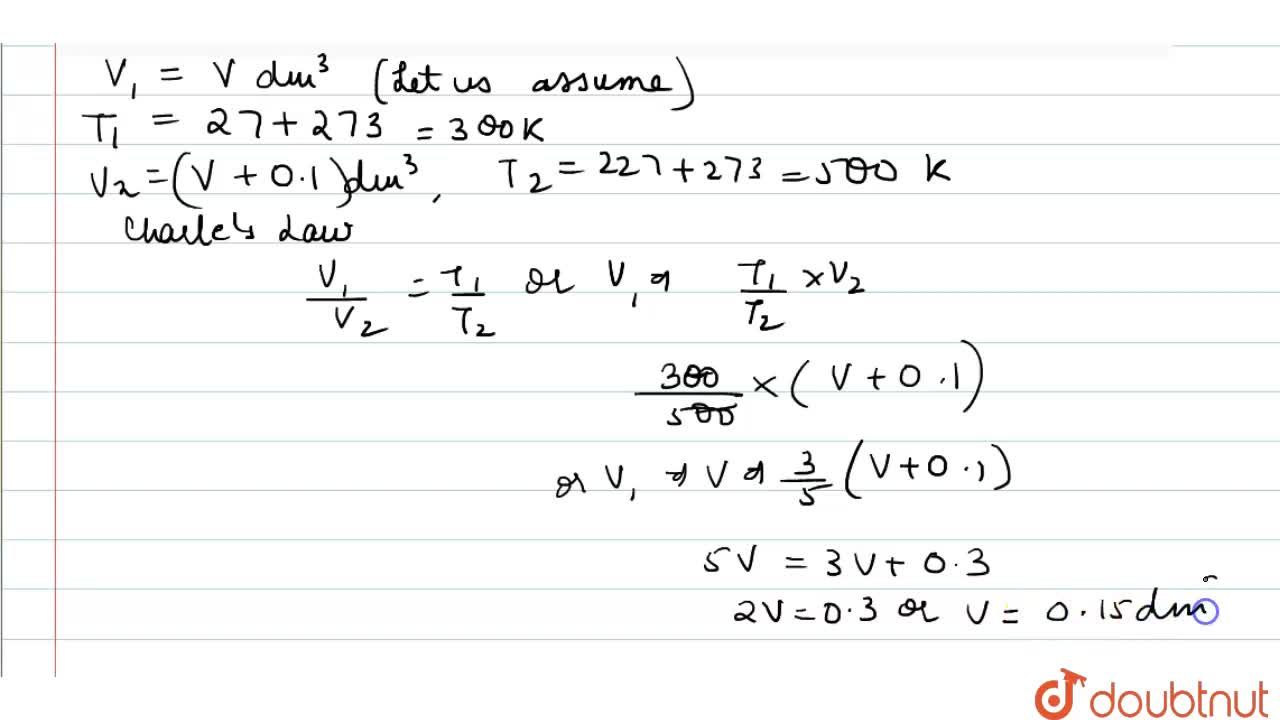




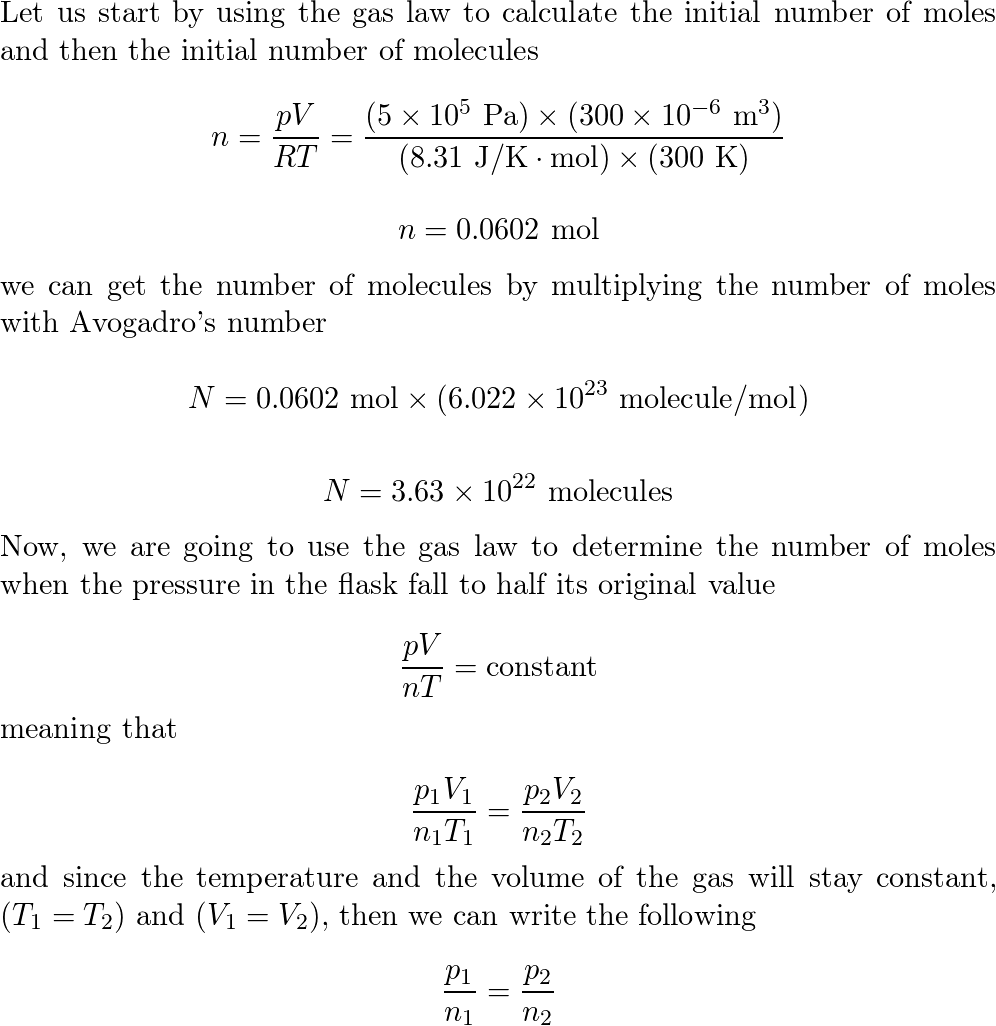

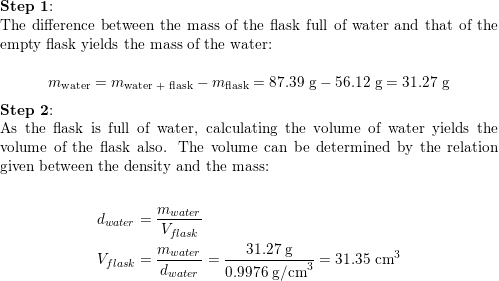
:max_bytes(150000):strip_icc()/GettyImages-493151728-c08e9b2d60bb401b8bbdd2e66de7a93b.jpg)