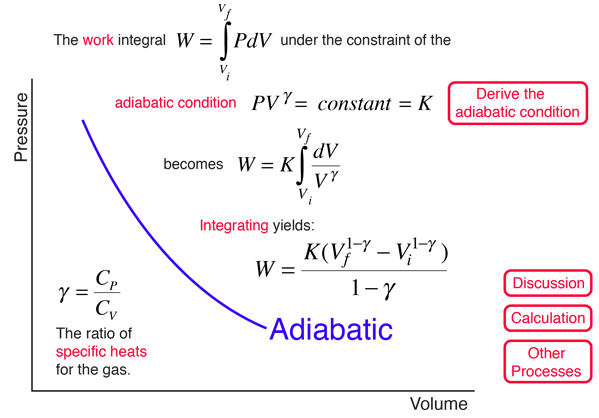
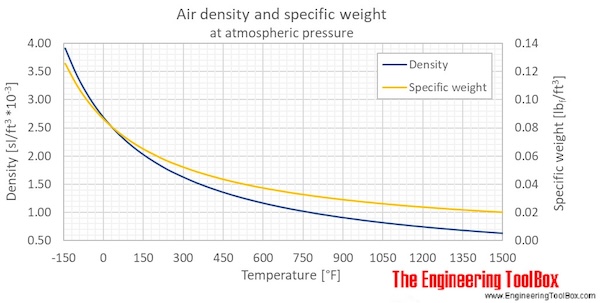
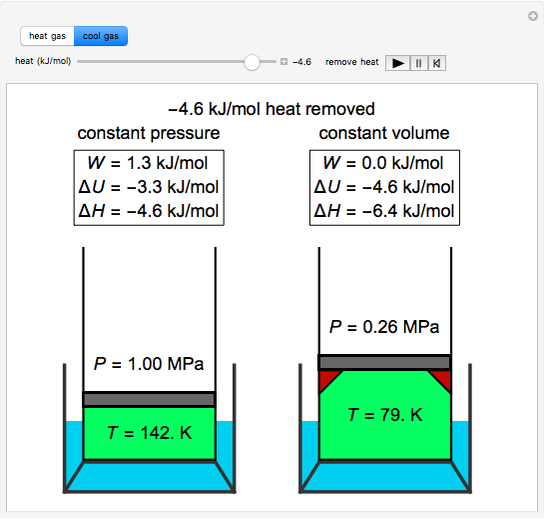
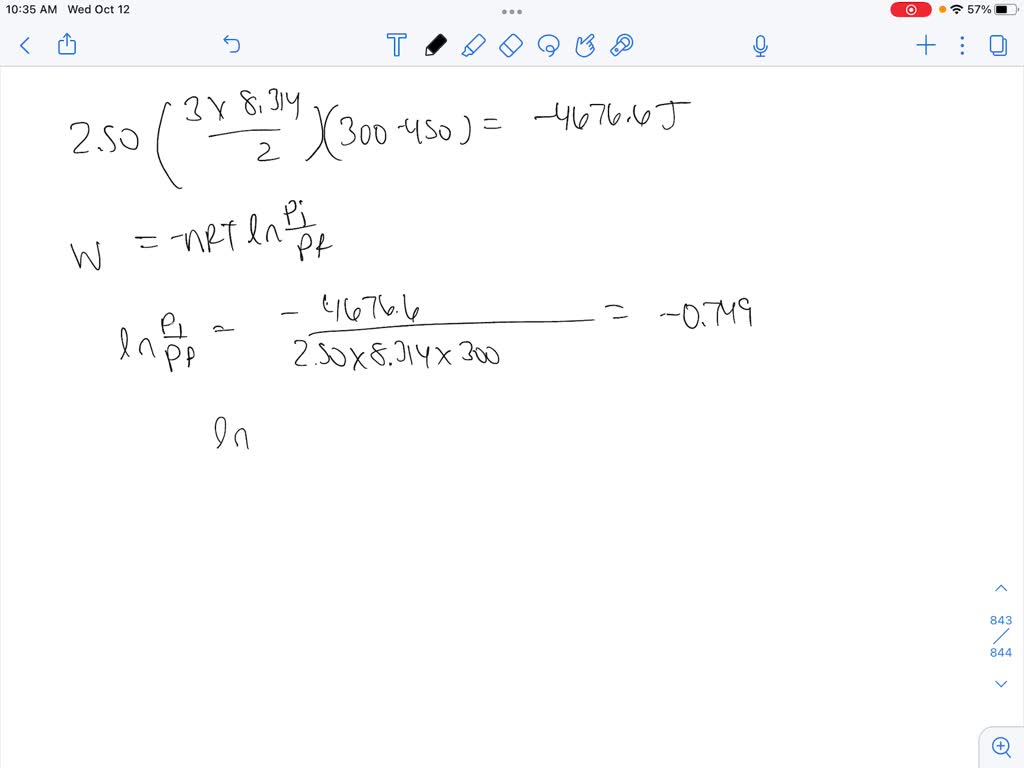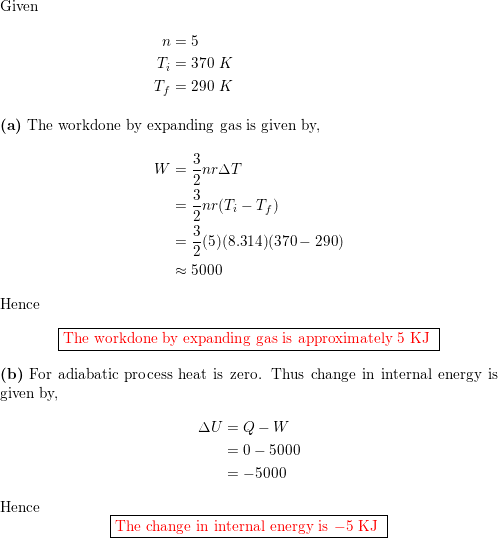A gas expands from `3 dm^(3)` to `5 dm^(3)` against a constant pressure of 3 atm. The work done - YouTube

Fig. S 6: Gas expansion factor as a function of calculated centreline... | Download Scientific Diagram

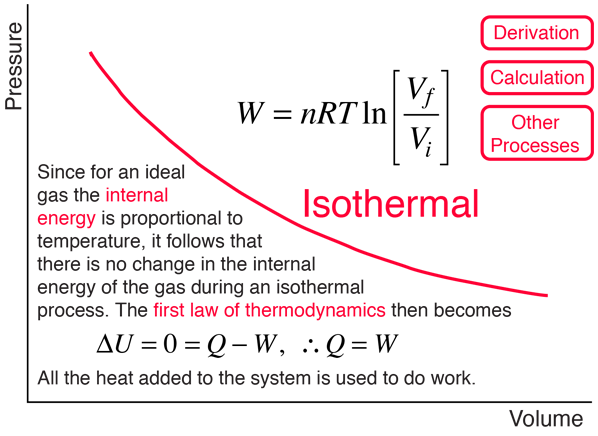
SOLVED: 3A) One (1) mole of an ideal gas receives an input of 360J of energy in the form of heat: The gas does 325J of work on its surroundings by expanding; (

How is the final temperature of irreversible adiabatic expansion of an ideal gas calculator? - Quora

SOLVED:A sample of nitrogen gas expands in volume from 1.6 to 5.4 L at constant temperature. Calculate the work done in joules if the gas expands (a) against a vacuum, (b) against

Methane Gas Volume Expansion Ratios and Ideal Gas Deviation Factors for the Deep-Water Bering Sea Basins: Peng-Robinson Equation of State