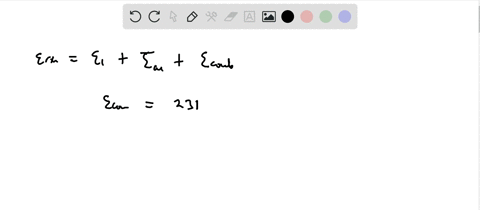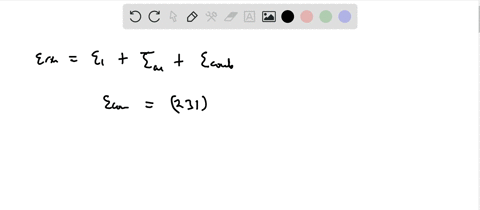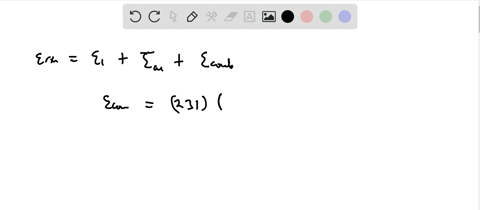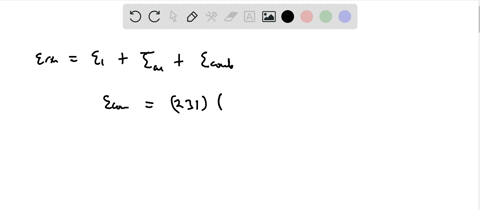
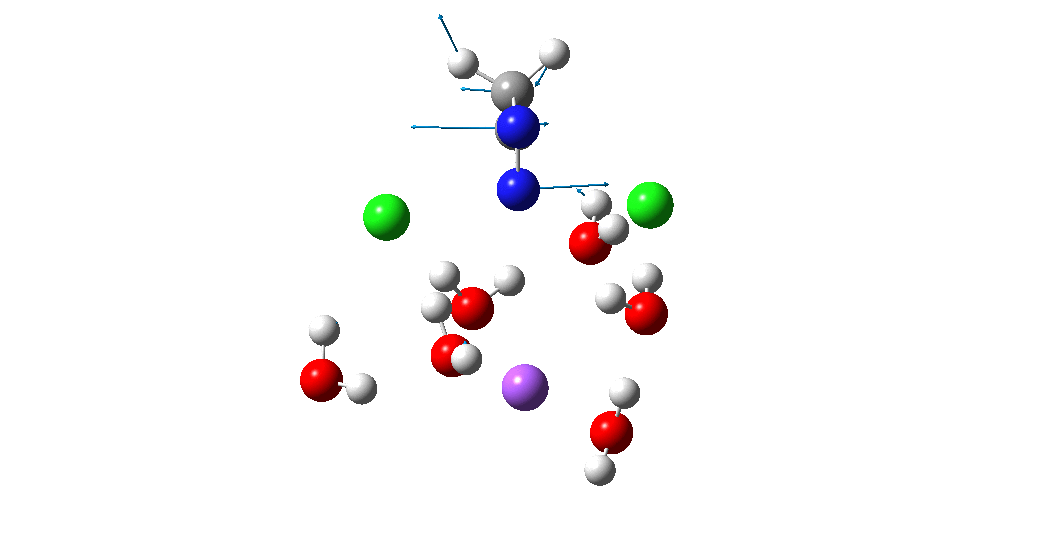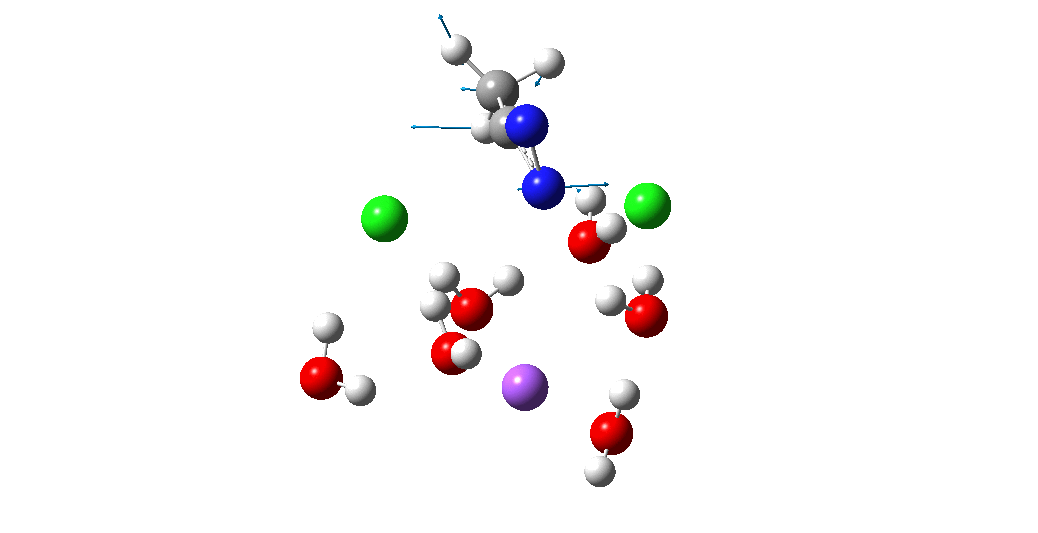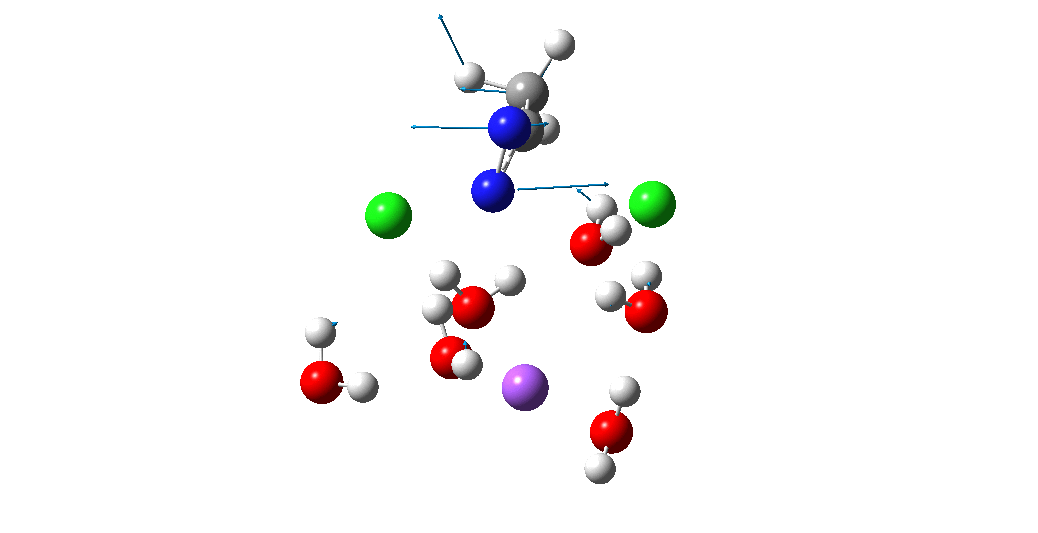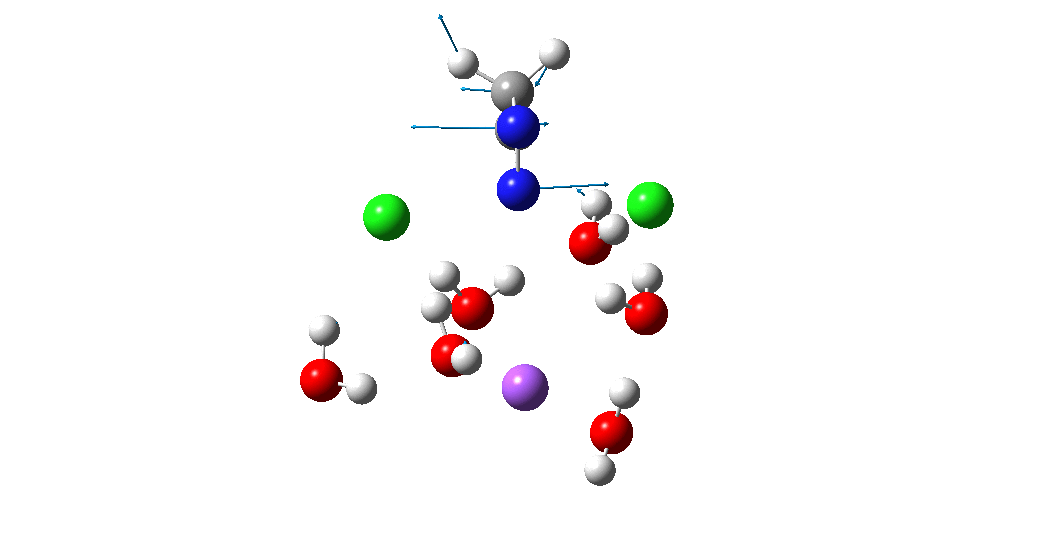
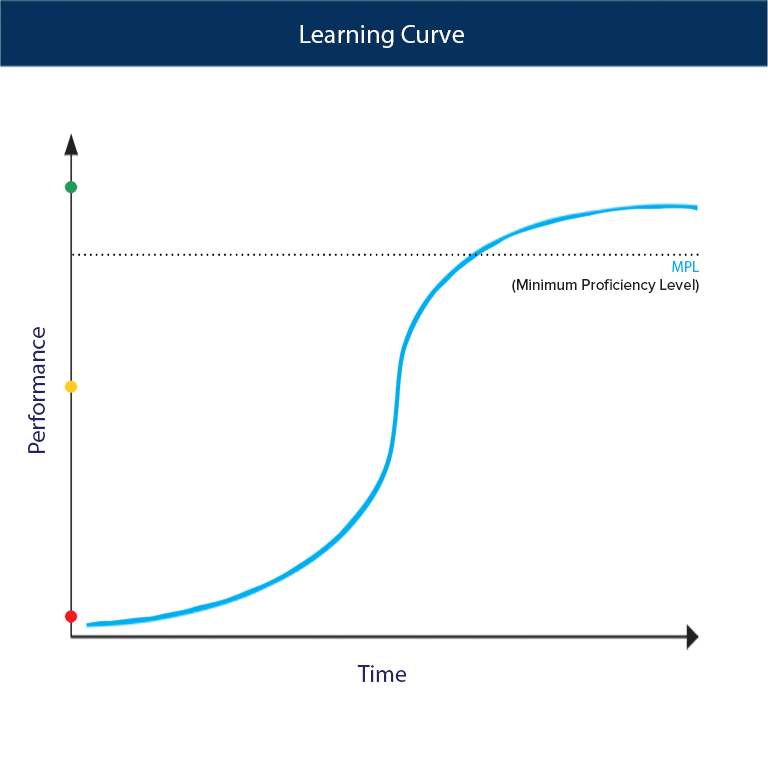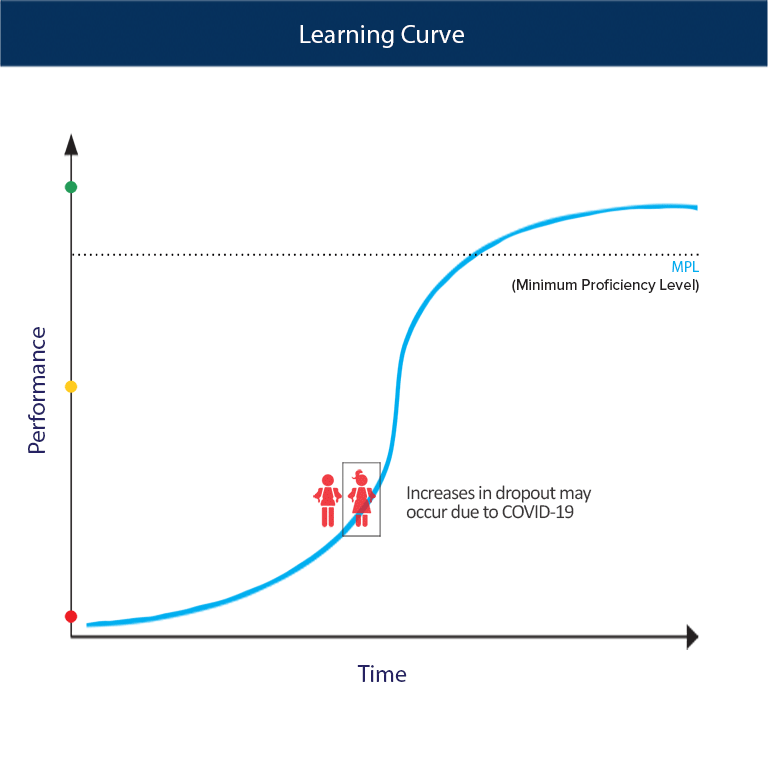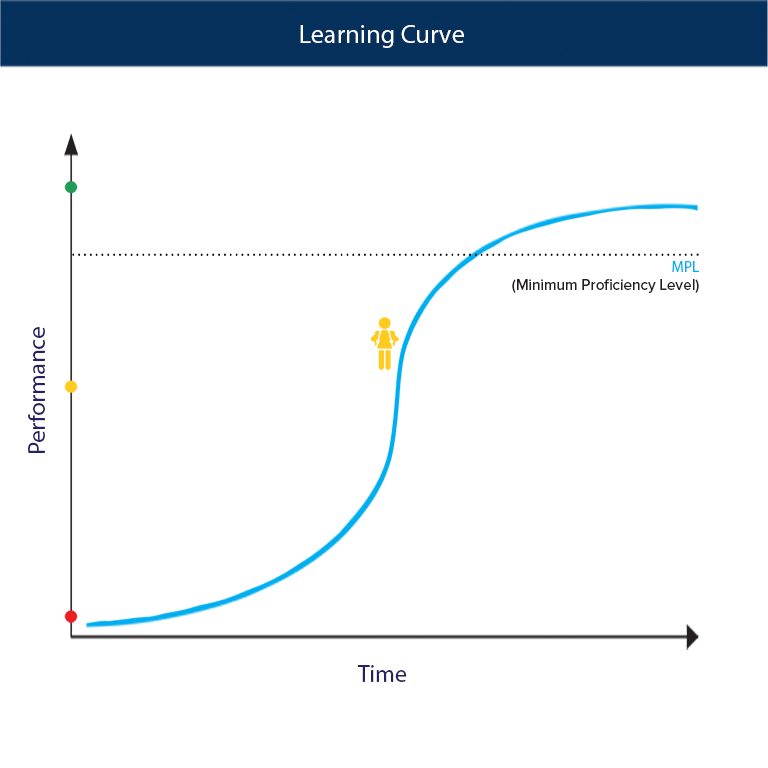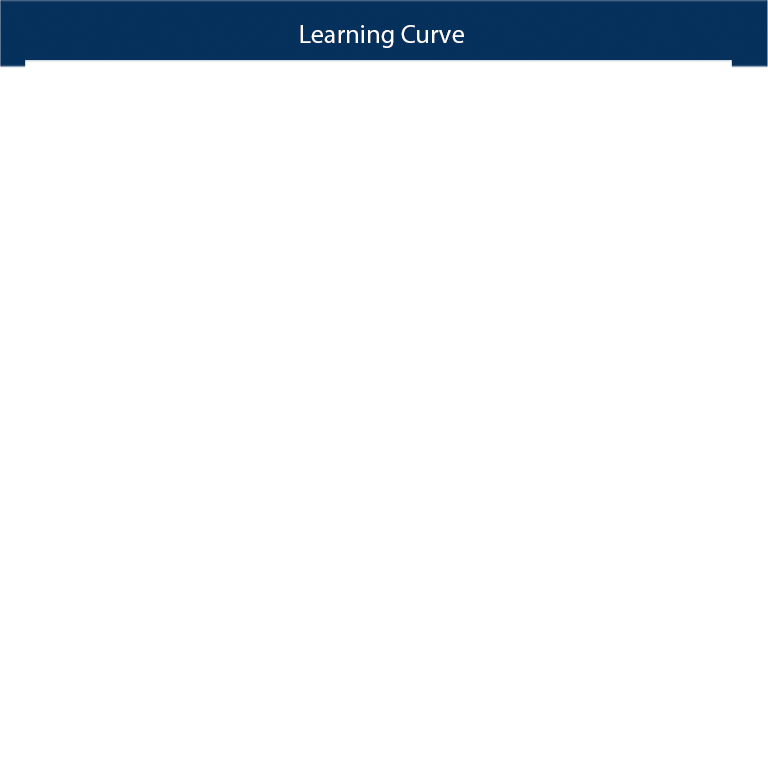
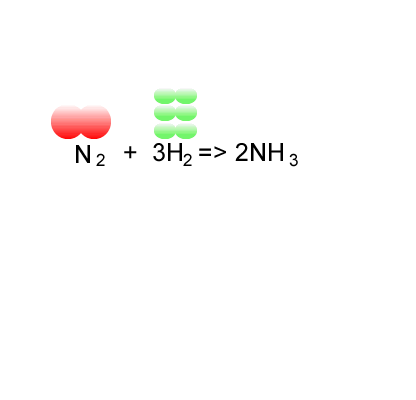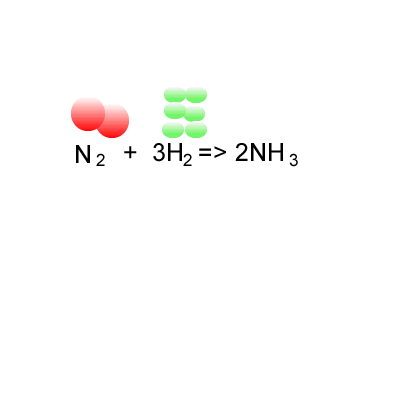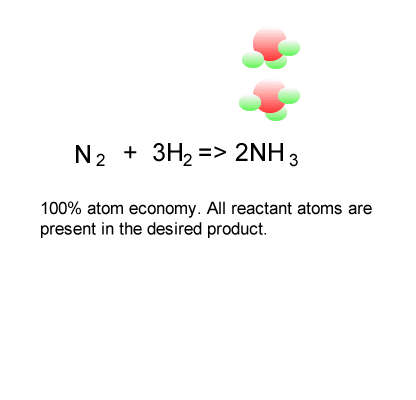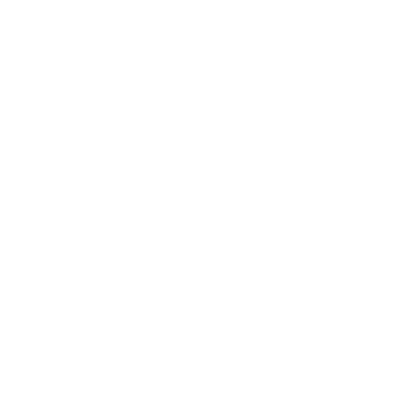
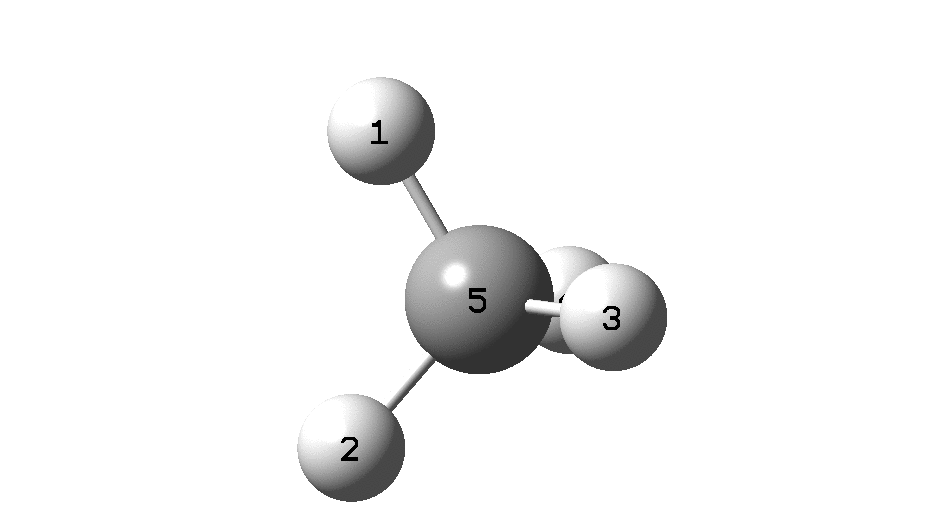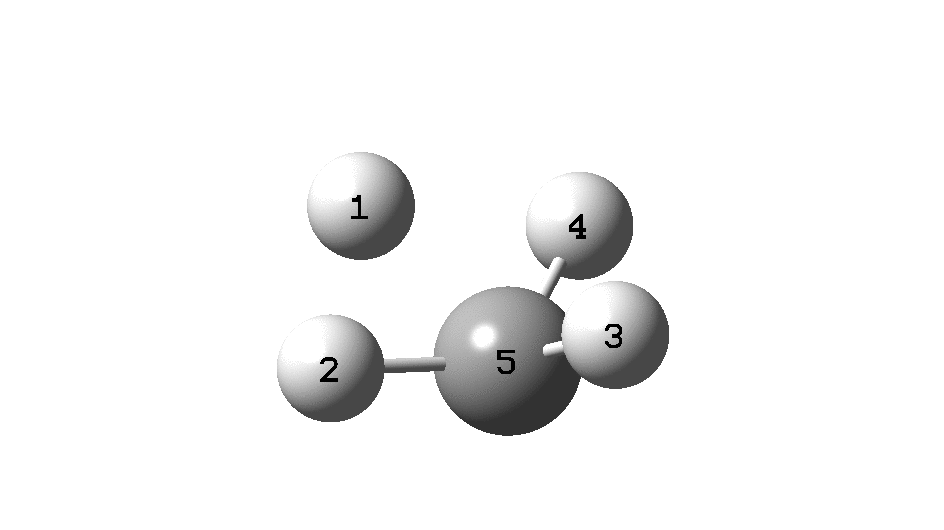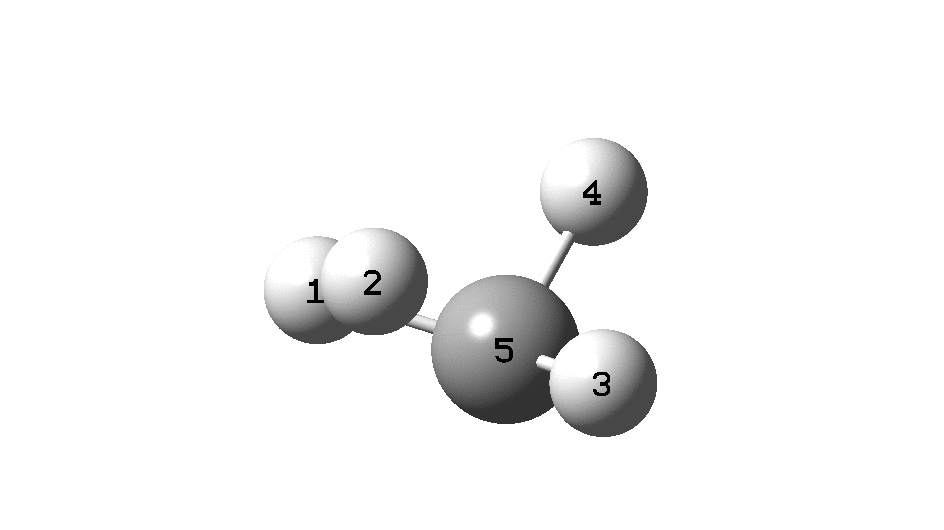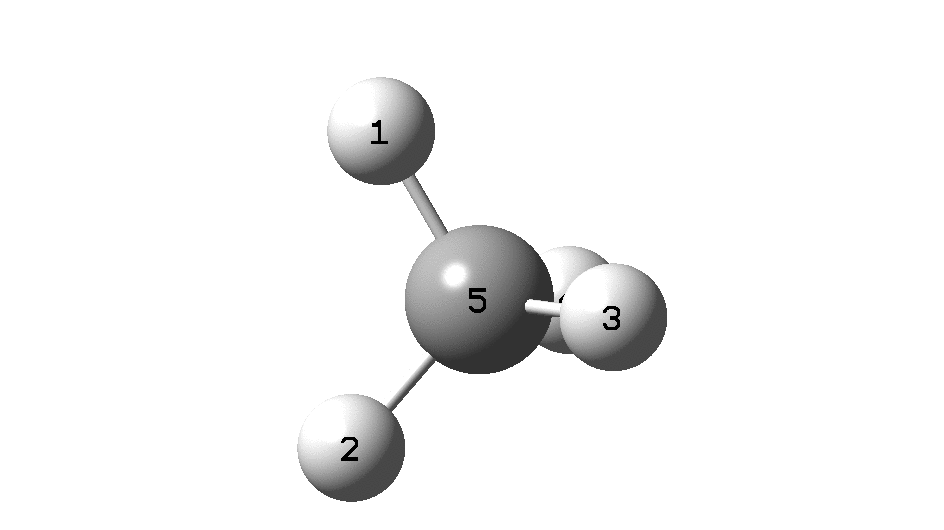Calculation of the activation barrier for CO dissociation on Cobalt Fischer-Tropsch Catalysts | MATLANTIS

SOLVED: Calculate the value of ΔG at 700 K for the reaction n X ⟶m B .^-1 Given that value of ΔH=-113 kJ mol^-1 and ΔS=-145 JK mol^-1 .
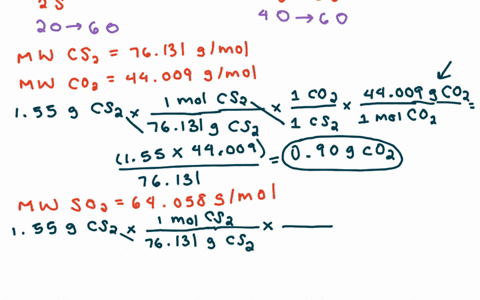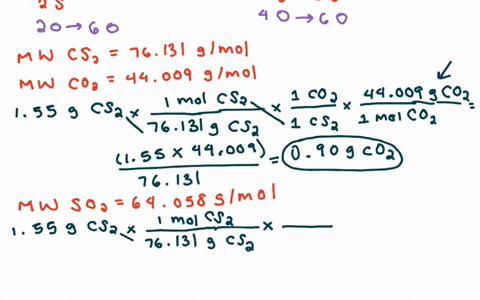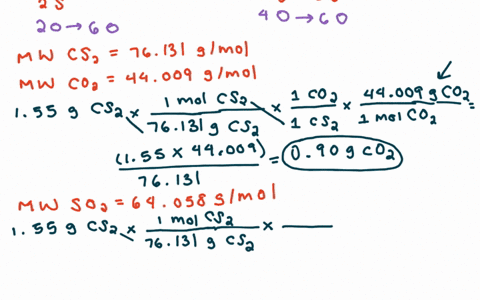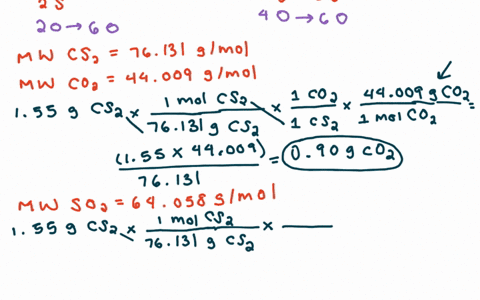
SOLVED:For each of the following unbalanced equations, calculate the mass of each product that could be produced by complete reaction of 1.55 g of the reactant indicated in boldface. a. 𝐂 𝐒2(l)+O2(g)