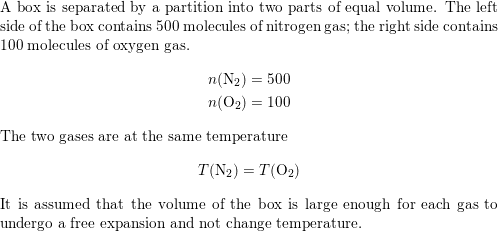Precision Nitrogen Oven/Precision N2 Box Oven, Precision Hot Air Oven Wholesaler - Precision Nitrogen Oven - Precision Nitrogen Oven

Two rigid boxes containing different ideal gases are placed on table. Box A contains one mole of nitrogen at temperature T0 , while box B contains 1 mole of helium at temperature
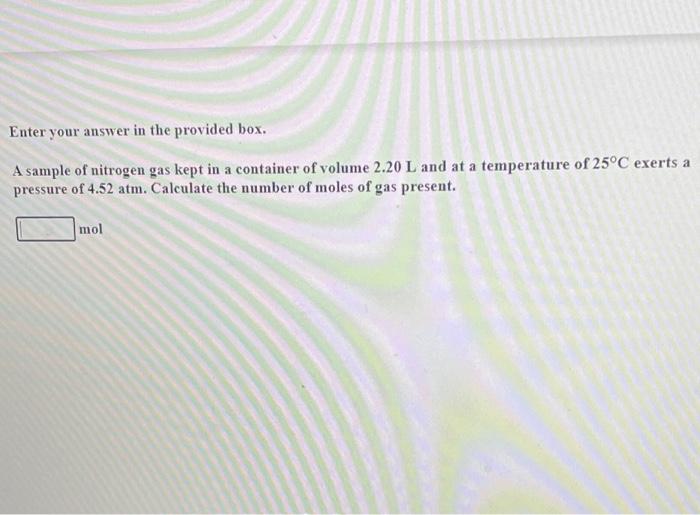
![Nitrogen gas is filled in a container of volume 2.32L at 32^oC and at 4.7 atm pressure. Calculate the number of moles of gas.[Write up to 3 decimal places] Nitrogen gas is filled in a container of volume 2.32L at 32^oC and at 4.7 atm pressure. Calculate the number of moles of gas.[Write up to 3 decimal places]](https://dwes9vv9u0550.cloudfront.net/images/4730475/db9edf02-73d5-4fdb-b5b6-bd216403235a.jpg)
Nitrogen gas is filled in a container of volume 2.32L at 32^oC and at 4.7 atm pressure. Calculate the number of moles of gas.[Write up to 3 decimal places]

SOLVED: 20.34 . A box is separated by a partition into two parts of equal volume. The left side of the box contains 500 molecules of nitrogen gas; the right side contains
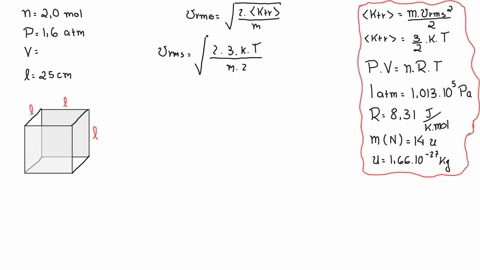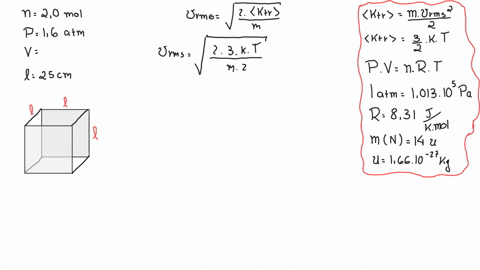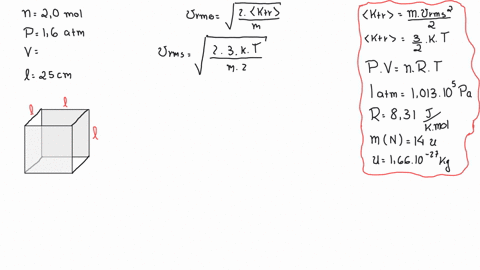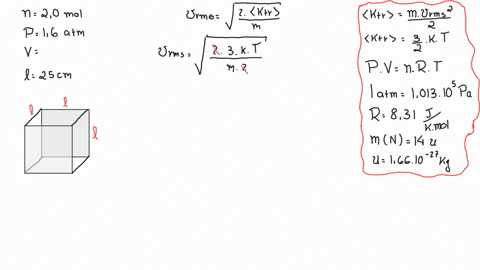
SOLVED: A cubic box of volume 5.1 x10^-2 m^3 is filled with nitrogen gas at atmospheric pressure (1.013x 10^5 Pa) and temperature equal to 40 degrees C. What is the root mean